 All the isotopes of Fr have very short half-lives, in contrast to the other elements in group 1.Įquation 21.6 2RbOH(s) + Mg(s) → 2Rb(l) + Mg(OH) 2(s) 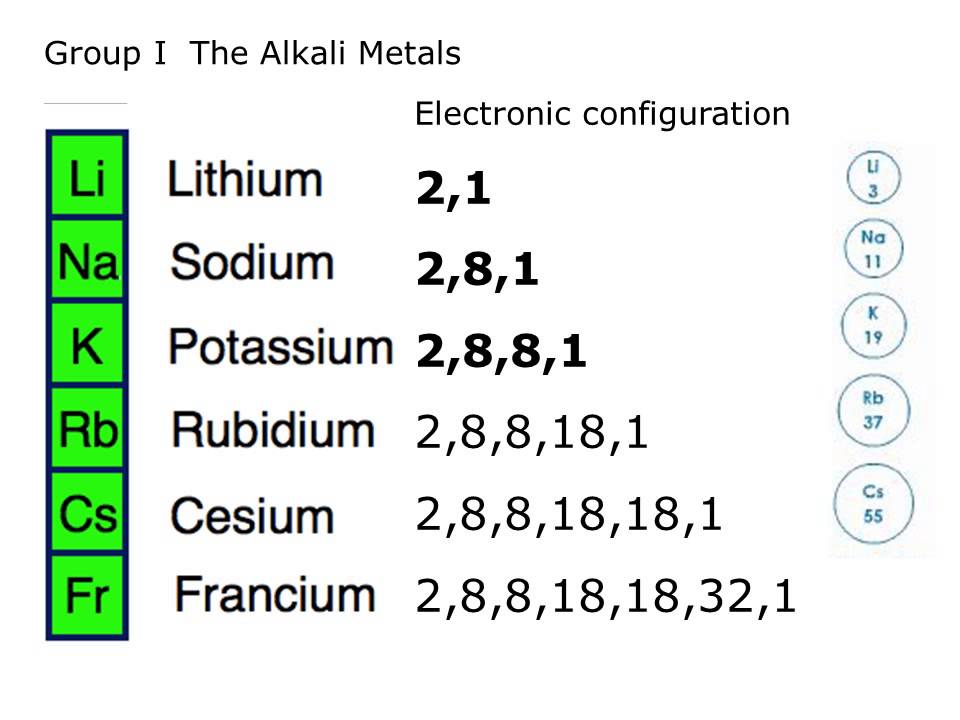
Francium (Fr) is found in only trace amounts in nature, so our knowledge of its chemistry is limited. Known to chemistry students as the inventor of the Bunsen burner, Bunsen’s spectroscopic studies of ores showed sky blue and deep red emission lines that he attributed to two new elements, Cs and Rb, respectively. Cesium (Cs) and rubidium (Rb) were not discovered until the 1860s, when Robert Bunsen conducted a systematic search for new elements. (The ashes produced by the combustion of wood are largely composed of potassium and sodium carbonate.) Lithium (Li) was discovered 10 years later when the Swedish chemist Johan Arfwedson was studying the composition of a new Brazilian mineral. For these reasons, the group 1 elements were unknown until the early 19th century, when Sir Humphry Davy first prepared sodium (Na) and potassium (K) by passing an electric current through molten alkalis. Although some of their ores are abundant, isolating them from their ores is somewhat difficult. The alkali metals are so reactive that they are never found in nature in elemental form. 
To be familiar with the reactions, compounds, and complexes of the alkali metals.To describe how the alkali metals are isolated.zip file containing this book to use offline, simply click here. You can browse or download additional books there. More information is available on this project's attribution page.įor more information on the source of this book, or why it is available for free, please see the project's home page. 
Additionally, per the publisher's request, their name has been removed in some passages. However, the publisher has asked for the customary Creative Commons attribution to the original publisher, authors, title, and book URI to be removed. Normally, the author and publisher would be credited here. This content was accessible as of December 29, 2012, and it was downloaded then by Andy Schmitz in an effort to preserve the availability of this book. See the license for more details, but that basically means you can share this book as long as you credit the author (but see below), don't make money from it, and do make it available to everyone else under the same terms. This book is licensed under a Creative Commons by-nc-sa 3.0 license. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
